Back to Tenrec Resources and Information
Tenrecs and solenodons in captivity
John F. Eisenberg
Resident Scientist, National Zoological Park, Smithsonian Institution, Washington, DC 20009, USA
From: International Zoo Yearbook 15 (1975): pp 6-12. Published by the Zoological Society of London.
Online publishing with kind permission of the International Zoo Yearbook and the Zoological Society of London.
© International Zoo Yearbook / Zoological Society of London. Online conversion by David Kupitz.
The living zalambdodont insectivores include families: Chrysochloridae, Tenrecidae, Potamogalidae, and Solenodontidae. These morphologically conservative mammals have received increasing attention during the last ten years (17). Some of the genera, like the solenodons, are extremely rare and in danger of extinction, while others, because of their physiological and reproductive peculiarities have been studied intensively (9). In this paper I will discuss our experiences with only the solenodons and tenrecs.
The solenodon was the subject of an earlier monograph by the late Dr E. Mohr (14). Our early experiences with solenodons, including aspects of their behaviour and their production of ultrasonic signals, have been published (3). Information on the husbandry of tenrecoid insectivores in captivity is included in previous publications (8; 10; 4; 7; 6). Rather than reiterate previously published material, we will discuss our experience in long term captive management and add new data acquired since then.
Solenodon paradoxurus in captivity
We imported five adult and three juvenile solenodons between 1965 and 1967. None of the adults survived more than 14 months in captivity. Various problems contributed to their premature deaths, in particular, stress and in at least three cases our inability to successfully convert them to an artificial diet. Of the three juveniles, one lived three years 73 days, the second five years seven months, and the third is still living at the time of writing. This animal has lived for nine years six months in captivity and is thought to be nearly ten years old.
Each animal of our longest surviving pair was fed the following daily: 3/4 cup of finely ground lean horsemeat plus 1/4 teaspoon of powdered milk, 1 raw egg yolk, 1 teaspoon of finely ground dry dog food, and 1 pinch of iodized salt. The mixture was placed in a shallow pan to prevent the animal from spreading it on the floor. The diet was supplemented with earthworms, crickets, hairless newborn mice, and two- to three-day old chicks, when available. Water was always on hand.
This pair was maintained together periodically in an enclosure measuring 3.6 x 2.4 m. The smooth wood sides of the enclosure prevented climbing. Solenodons do attempt to climb vertical surfaces and we wanted to prevent the possibility of an animal falling and injuring its nose or limbs. They have a habit of putting their snouts into cracks and crevices, so it was also necessary that the wall be free of cracks. The wood floor of the cage was covered with newspapers or an earth-humus mixture. Rotten logs or hollow logs were occasionally placed in the enclosure which helped prevent the development of stereotyped locomotion patterns. The logs or branches were devoid of sharp projections or cracks in which the solenodon could catch its tail, foot or snout.
The animals were provided with an L-shaped nestbox with an entrance 15 cm wide and 20 cm high. The entrance tunnel was 30.5 cm long and opened into a nest chamber 30 x 46 x 46 cm, provided with newspaper strips for nesting material. The animals were kept at 21-27°C and we attempted to maintain a high humidity of around 50%.
It is imperative that the axillary areas, abdomen and tail as well as the soles of the feet be frequently inspected since a solenodon tends to develop dry patches on the skin. If slight abrasions are found, they should be treated with A-D ointment, as they may be scratched until an open sore develops. Similar problems were described by Mohr (14).
From studying the age structures of family groups captured in Hispaniola as described by Mohr (14), I suggest that solenodons would do best if kept in twos - a natural family group consisting of a mother and her offspring - or alone. Maintaining groups of adults of both sexs undoubtedly creates an unnatural stress situation.
Since 1966 we have observed courtship, mounts, and attempted intromissions, but no fertile mating has resulted. Utilizing a vaginal smear technique, it was determined (by R. Alisio) that oestrus lasts less than a day; the interval between oestrous periods in a given female ranges from nine to 13 days. Mating behaviour follows a typical insectivore pattern: an adult male, when introduced into a female's cage, will generally explore and scent-mark by dragging the anogenital area over protuberances in the cage. He often uses areas which have been scent-marked previously by the female. The male usually urinates and defaecates at the same spot in the female's cage. Initial contact between the pair involves mutual nose to nose contact or nose to body contact. Sniffing at the neck, ear, flank, anogenital, chest, and side are frequent. During the initial encounter, the 'piff' sound is repeated at intervals until the animals come together. It would appear that this short note is a contact sound allowing the animals to locate one another. The female has been seen attempting to mount the male when she is in oestrous. When the male mounts, he employs a neck grip to orientate himself and to hold the female; although attempted intromissions were noted, complete copulation was never observed.
A lactating female and her infant judged, according to the criteria given by Mohr, to be some 50 days of age were imported. We observed a unique mode of maternal-young contact which we have referred to as 'teat transport'. This phenomenon is well known among some rodents (2), but unreported in insectivores. At seven weeks of age the youngster will accompany its mother on her foraging activities by clinging to one of the two inguinal teats. At this time the teats are very elongated, up to 2 cm in length, enabling the youngster to cling to a teat as it is dragged along close behind. As the infant grows it is able to assert its own locomotion and simply seize the teat and follow, moving when the mother moves, stopping when she stops. The 21/2 month old infant may still show this response and even scratch itself while standing behind the mother holding on to a teat. It would seem that if the solenodon has to change burrows from time to time, then such a teat-transport mechanism enables the female to move with still very dependent young, pulling them along behind her on her teats, rather than attempting to carry them in her mouth. Mouth transport, of course, is a wide-spread phenomenon in small insectivores; however, the solenodon's teat-transport mechanism is probably quite efficient since the young remain dependent for a long time. During this dependency period, the female can forage and be accompanied by the young. If Mohr's growth curve (14) is accurate and our estimate of the infant's age at arrival is correct, then this dependency phase lasts some 21/2 months.
Solenodon typically produces one or two young and Mohr (14) estimated their birth weight at approximately 100 g. Judging from the data on tenrec gestation, the gestation period of Solenodon may be in excess of 50 days. If lactation lasts for some 75 days and induces a lactation anoestrous, the interval between births must be about 125-135 days. If a female averages two young per litter, then she would not produce more than four young per year. The solenodon would seem to exhibit a pattern of very low fecundity and a rather long life span. The same also applies to the tenrec genus Microgale (6).
The Tenrecidae
Information concerning tenrecoid insectivores in captivity has been summarized elsewhere (5; 6). Colonies of tenrecs have bred successfully in many zoos and the utility of Echinops telfairi as a laboratory animal has already been discussed (8). A detailed study of captive propagation was published by Honegger and Noth (10) and breeding into the fourth and fifth generations is well established in several collections (16). Our original Setifer setosus stock was sent to the Jersey Zoo where it has since flourished (16).
Exhibition: With the exception of Hemicentetes semispinosus, most tenrec species are nocturnal and for this reason must be exhibited under reversed lighting, generally under red light. Successful exhibits of Hemicentetes were set up in the Bird House at the National Zoo and in the Small Mammal House at the New York (Bronx) Zoo.
Survival rate and fertility: Although breeding was achieved to the third generation with H semispinosus, we were unable to consolidate our colony as we did not fully understand their life cycles and fertility. Fig. 1 presents a survival curve for this species and for the animals which remained in our collection until their death. Starting with the five original wild-caught adults, we see that the maximum longevity attained was 26 months. Assuming that they were several months old at the time of arrival, the longevity compares favourably with that noted for captive-born specimens. Of 36 captive-born animals, the life expectancy shows an initial mortality during the first month which stabilized until about one year of age. From then on, there is a decline, all 36 captive-born animals having died by 30 months 2 weeks of age. Such information is invaluable when planning a breeding programme.
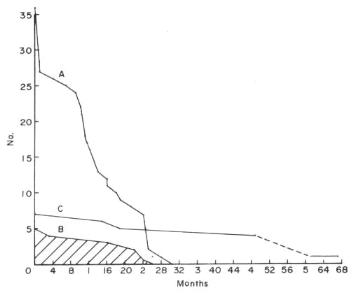 Fig. 1 Survival rate of five wild-caught (B) Hemicentetes semispinosus compared with that of 36 captive-born (A) specimens. Seven wild-caught Microgale talazaci (C) achieved a maximum longevity of 51/2 years.
Fig. 1 Survival rate of five wild-caught (B) Hemicentetes semispinosus compared with that of 36 captive-born (A) specimens. Seven wild-caught Microgale talazaci (C) achieved a maximum longevity of 51/2 years.
Click here or on the figure to view a larger version.
When H. semispinosus began to breed we assumed that the animal's fertility would continue over a period of at least two years and we overestimated their potential longevity. As a result we deferred breeding during the next two years because we had no space for keeping large numbers. We should have transferred more animals to other institutions for our delay in breeding them resulted in lower production, decreased fertility and eventually the loss of the entire colony.
An examination of the survival rate of Microgale talazaci presents an entirely different picture. Starting with our original seven animals, survival was excellent with only three dying before they were four years old (Fig. 1). For such a small mammal, a longevity of 51/2 years is excellent. Our previous publication (6) shows that the animals can reproduce well into their fourth year. The young are slow to mature and the litter size is small; hence, starting from a small stock, it is difficult to build up a large colony for a number of years. Note again, the correlation between low litter size and long breeding age, as was inferred for Solenodon. The same is probably true of the large gymnure Echinosorex gymnura, since this species has only one to two young per litter and apparently survives in captivity as long as seven years (11).
Reproductive biology: Mating behaviour has been described in some detail in a previous piblication (5). Unlike the Common house shrew Suncus murinus (1), tenrecs have rather extended mounts when copulating. In Suncus, the male makes a series of brief mounts preceeding a terminal mount with ejaculation, averaging about 38 mounts with intromission prior to an ejaculatory mount (1). Following insemination the female house shrew will generally behave aggressively toward the male and subsequent matings do not take place.
Unlike Suncus, the male tenrec courts a female and attempts to mount even if she is not completely receptive. Mounts with intromission are prolonged: 71/2 minutes for Microgale dobsoni; 41/2-12 minutes for Tenrec ecaudatus; approximately 10-21 minutes for H. semispinosus, over 28 minutes for S. setosus; and 16 to over 18 minutes for E. telfairi. It is not known how many times the male tenrec ejaculates during a mount; however, the male may continue to court the female and mount her on subsequent days. Gradually his attentions wane and mating activity ceases.
Since, in most breeding attempts, we have not separated a male immediately after a long mount but instead have left him with the female for up to four days, we are not certain when conception takes place. We determined gestation from the time of the first copulatory mount series until the birth of the young but now realize that tenrec females may be induced ovulators and conception may not take place during the first mating session. If this is so, then our recorded gestations may be somewhat longer than the true gestation period (from actual fertilization to the birth of the young).
Another complicating feature in the determination of accurate gestations is that tenrecs are capable of showing some true variation in length of gestation. This may be related to litter size and may also be related to the average ambient temperature to which the pregnant female is subjected. More refined studies will no doubt provide an accurate gestation length for the Tenrecidae but at the present time, it seems worthwile summarizing what we know. Mallinson gives a range of 61-64 days for gestation in E. telfairi (13). This compares rather favourably with our data (7) for Echinops of 62-65 days. Prior to that Eisenberg & Gould had listed a gestation of 68 days or under (4).
For S. setosus, Eisenberg & Muckenhirn (7) recorded from 65-69 days, based on first observed copulations, as the interval for gestation. Mallinson reports 54-61 as the range at the Jersey Zoo with the mode occurring at 56 days (13). This may be attributable to the higher ambient temperature at which they maintain female Setifer, but it may also indicate that several copulations are necessary over several days before fertilization takes place.
H. semispinosus appears to have a gestation of between 55 and 57 days, as does H. nigriceps. T. ecaudatus has a slightly longer gestation period, more than 58 and less than 64 days. Microgale species were reported by Eisenberg & Maliniak to have gestation periods of about 62 days (6); however, in the last year we have determined that they may be as low as 58 days. In spite of some possible errors, the gestation for most of the tenrecoid insectivores appears to lie between eight and nine weeks (Table 1).
It is generally agreed that Hemicentetes and Tenrec are closely related genera (5). They have gestation periods which are nearly the same, in spite of the fact that a female Tenrec may be five times heavier than a female Hemicentetes. It would appear, however, that Tenrec has on the average a larger litter than Hemicentetes.
| |
Maximum
captive
longevity
(yrs mths) |
Litter size
(range and
average) |
Recorded
gestations
(days) |
Birth weight
of young
(g) |
| Solenodontidae |
|
|
|
|
| Solenodon paradoxurus |
9 6 |
1-2 |
-- |
c. 100 |
| Tenrecidae |
|
|
|
|
| Tenrecinae |
|
|
|
|
| Tenrec ecaudatus |
4 5 |
1-32 (c. 15) |
57 <= 63 |
25 |
| Hemicentetes nigriceps |
2 7 |
2-4 (2.8) |
>= 55 |
6.3 |
| Hemicentetes semispinosus |
2 6 |
2-11 (6.6) |
57 <= 63 |
6.5 |
| Setifer setosus |
4 7 |
1-5 (3) |
65 <= 69 |
24.7 |
| Echinops telfairi |
6 |
1-10 (7) |
62 <= 65 |
c. 6.0 |
| Oryzorictinae |
|
|
|
|
| Microgale (Nesogale) talazaci |
5 6 |
1-3 (2) |
>= 58 <= 63 |
3.6 |
| Microgale (Nesogale) dobsoni |
5 7 |
1-2 |
62 <= 64 |
c. 4.0 |
Table 1. Solenodons and tenrecs kept at the National Zoological Park, Washington.
Development of the young: One way to assess development of the young and the state of altriciality in the young is to add the gestation period to the time it takes for the youngster's eyes to open. We then see that the extremes for the Tenrecidae are 65 and 80 days (6; 7) with a mean for most species of between 70 and 75 days. H. semispinosus shows the shortest time for total development at about 65 days, whereas M. talazaci shows the longest from about 75 to 80 days.
The degree of altriciality of the young is roughly indicated by the ratio between the weight of the young at birth divided by the average weight of the adult female. T. ecaudatus young are the most altricial at birth, whereas the other species are roughly at a comparable stage of development. Extending the comparisons to S. paradoxurus based on (14), Solenodon is seen to produce the most precocial young. Nevertheless, all zalambdodont insectivore young are born with auditory openings and eyes closed, and only the sparsest appearance of hair or spines.
Maternal care patterns for the tenrecs are described in Eisenberg & Gould (5). All species build a nest in a secluded place, nestbox or burrow. Nursing and maternal brooding continue in the nest until the young are developed sufficiently to begin following the mother on her nightly excursions. Of course, during the early period of development, the female will leave the nest to forage independently, returning to nurse the young. All tenrecoid insectivore females show a well developed retrieving response; if a young should stray from the nest, it will be picked up in the mouther's mouth and carried back to the nest site.
Social groups: Social tolerance among adults is highly variable. There is some evidence that M. talazaci develops a pair preference (6). Tenrec, Setifer, and Echinops females with litters should be isolated, although cases of male tolerance have been recorded for Setifer and Echinops. H. semispinosus exhibits the greatest social tolerance, although we have had the best rearing success when the pregnant female is isolated before giving birth. Nevertheless, Hemicentetes may form small family groups of several generations using an extended burrow system (5).
Conclusions
The captive maintenance of insectivores presents many challenges. Considerable success has been achieved with E. telfairi (8; 10; 16) and Setifer (16). H. semispinosus with its early maturation and short potential life span presents certain problems. A successful breeding programme can be designed for this species if its short period of fertility is taken into consideration. T. ecaudatus has had considerable success at Wassenaar Zoo (12), but it is not an easy species to manage on a long term basis. The various Microgale species that we studied have proved to be long-lived, remarkable animals and we feel optimistic about the prospects of long-term breeding in captivity.
Solenodon is difficult to acclimatize. Although we have had remarkable longevity records with animals taken as juveniles, we strongly discourage the keeping of wild-caught adults in captivity until certain, as yet unknown, requirements can be met during acclimatization. If further research on acclimatization or breeding is to be undertaken it should be carried out in Hispaniola itself in co-operation with the local authorities. Full protection has been given to S. paradoxurus in the Dominican Republic as well as for S. cubanus on Cuba (15).
Product mentioned in text
A-D ointment: manufactured by White Laboratories, Kenilworth, New Jersey, USA.
References
1. DRYDEN, G. L. (1969): Reproduction in Suncus murinus. J. Reprod. Fert. (suppl.) 6: 377-396.
2. EIBL-EIBESFELDT, I. (1958): Das Verhalten der Nagetiere. Handbuch der Zoologie VIII, 10 (13): 1-88.
3. EISENBERG, J. F. & GOULD, E. (1966): The behaviour of Solenodon paradoxurus in captivity with comments on the behavior of other Insectivora. Zoologica, N.Y. 51: 49-58.
4. EISENBERG, J. F. & GOULD, E. (1967): Maintenance of tenrecoid insectivores in captivity. Int. Zoo Yb. 7: 194-196. (Free Full Text)
5. EISENBERG, F. F. & GOULD, E. (1970): The tenrecs: a study in mammalian behaviour and evolution. Smiths. Contr. Zool. 27: 1-137. (Free Full Text) (Alternative Free Full Text)
6. EISENBERG, J. F. & MALINIAK, E. (1974): The reproduction of the genus Microgale in captivity. Int. Zoo. Yb. 14: 108-110. (Free Full Text)
7. EISENBERG, J. F. & MUCKENHIRN, N. (1968): The reproduction and rearing of tenrecoid insectivores in captivity. Int. Zoo Yb. 9: 106-110. (Free Full Text)
8. GOULD, E. & EISENBERG, J. F. (1966): Notes on the biology of the Tenrecidae. J. Mammal. 47: 660-686. (Free Full Text)
9. HERTER, K. (1962): Untersuchungen an lebenden Borstenigeln (Tenrecinae), 1. Zool. Beitr. 7: 239-292.
10. HONEGGER, R. E. & NOTH, W. (1966): Beobachtungen bei der Aufzucht von Igeltanreks Echinops telfairi Martin. Zool. Beitr. 12: 191-218.
11. LIM, B. L. (1967): Note on the food habits of Ptilocercus gymnurus (Raffles) (Moonrat) in Malaya with remarks on 'ecological labelling' by parasite patterns. J. Zool., London 152: 375-379.
12. LOUWMAN, J. W. W. (1973): Breeding the tailless tenrec Tenrec ecaudatus at Wassenaar Zoo. Int. Zoo Yb. 13: 125-126. (Free Full Text)
13. MALLINSON, J. J. (1968): Establishing mammal gestation periods. Rep. Jersey Wildl. Preserv. Trust 9: 62-65.
14. MOHR, E. (1936-38): Biologische Beobachtungen an Solenodon paradoxurus in Gefangenschaft (Parts I-IV). Zool. Anz. 113: 177-188; 116: 65-76; 117: 233-241; 122: 132-143.
15. MORENO, A. (1966): La fauna de Cuba. III. Los Mamiferos. Zoologico 1 (5-6): 13-15.
16. RIORDAN, D. V. (1973): Reproduction in the spiny and pigmy hedgehog tenrec. Rep. Jersey Wildl. Preserv. Trust 9: 18-25.
17. VAN DE BROEK, G. (1961): The comparative anatomy of the teeth of lower and non specialized mammals. Intern. Colloq. on the Evolution of lower and non specialized Mammals Kon. VI. Akad. Wetensch. Lett. Sch. Kunsten Belgie, Brussels 1: 215-320.
Manuscript submitted 11 February 1974
 Back to the top
Back to the top
Back to Tenrec Resources and Information
 Fig. 1 Survival rate of five wild-caught (B) Hemicentetes semispinosus compared with that of 36 captive-born (A) specimens. Seven wild-caught Microgale talazaci (C) achieved a maximum longevity of 51/2 years.
Fig. 1 Survival rate of five wild-caught (B) Hemicentetes semispinosus compared with that of 36 captive-born (A) specimens. Seven wild-caught Microgale talazaci (C) achieved a maximum longevity of 51/2 years.